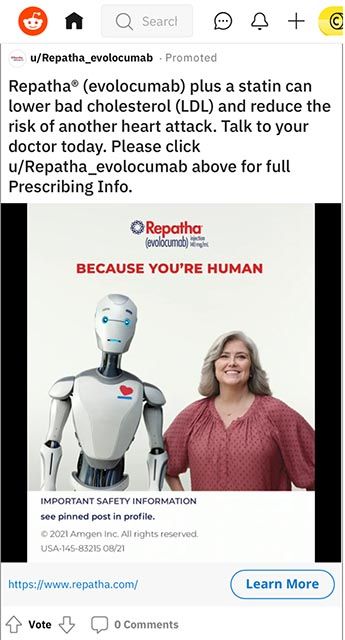
We frequently search for drug information across therapeutic categories, so naturally we get served drug ads everywhere we browse, including Reddit. We were recently served a drug ad on mobile Reddit that caught our eye—mostly for regulatory reasons. The most obvious missing piece we spotted was risk information, which would seem to run afoul of FDA fair balance requirements. However, FDA guidance is more lenient for ads with space/character constraints and its recommendations for internet/social media platforms contain more wiggle room than usual.2
Obviously this drug’s creative team felt confident including directions to view safety information hosted elsewhere, despite including a product claim that should technically be balanced by risk info. Their workaround here, which we’ve also seen on other platforms like Instagram, is pinning an Important Safety Information post in their user profile. The straightforward product claim does contain some indication info and is balanced with an eye-catching photo to make users pause in their endless scrolling.
It’s ultimately up to individual firms to decide on their risk tolerance in limited space/limited character count environments. We’ve worked with clients on both ends of the risk tolerance spectrum and know that good execution is down to more than just managing fair balance. With the right imagery, impactful language, and a regulatory-conscious creative process, it’s possible to make mobile work for you—even on Reddit.
References:
1Reddit User and Growth Stats – Updated October 2021. https://backlinko.com/reddit-users#why-do-people-use-reddit
2Guidance for Industry: Internet/Social Media Platforms with Character Space Limitations— Presenting Risk and Benefit Information for Prescription Drugs and Medical Devices. https://www.fda.gov/media/88551/download